Adsorption
Adsorption is a phenomenon of great industrial importance. Our laboratory develops research aimed at capturing CO2, energy storage, treating contaminated currents (H2S, dyes, toxins) and controlled drug release.
A method to increase the efficiency of molecular sieves to capture CO2 is through functionalization. Stronger adsorption sites are created in the adsorbent framework by impregnation with CO2 high affinity compounds. We have focused on the study of Molecular Organic Framework (MOFs) – Ionic liquids systems. We found that ionic liquids considerably extend the range of pressures in which the impregnation promotes increased in CO2 adsorption, unlike the zeolites, that were initially investigated using MEA. Recently, our studies with mesoporous MOFs showed that it is possible to double the CO2 adsorption capacity and extend the adsorption increment up to 2000 kPa, resulting in a record increase among the known functionalization techniques.
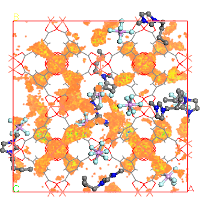 MOF Cu-BTC impregnated with ionic liquid
MOF Cu-BTC impregnated with ionic liquid
Conventional automotive natural gas (NG) storage technology is an obstacle to NG large-scale application. Conventionally the gas is compressed at 200 atm, the tank have very thick walls, its geometry is extremely limited and it is subject to explosion or fire in the event of a collision. With storage in an adsorbent bed, the pressure drops to 65 atm. The tank wall has a regular thickness, its geometry is free and the tank much safer. The testing of new adsorbent materials is extremely costly due to the number of cycles and the composition of the mixture of natural gas with more than five components. We use molecular simulation to assess the adsorptive capacity of materials such as MOFs and activated carbons, investigating relevant aspects in the choice of materials and operating conditions, such as the retention of larger molar mass compounds and delivery capacity of the adsorbent, including integrating with larger scale simulation of the adsorption process using a thermodynamic tank model.
 Adsorbed molar fraction of heavier hydrocarbons with the cycle number in maxsorb
Adsorbed molar fraction of heavier hydrocarbons with the cycle number in maxsorb
In recent years, the widespread use of biogas from landfills or waste water treatment plants, as source of clean and sustainable energy, there has been an increasing and urgent need to remove H2S from the biogas streams. The European Biogas Association (EVA) reports that, in the European Union countries, the number of biomethane plants grew from 187 plants in 2011 up to a total of 540 plants in 2018. We have investigated the role of ultramicroporous range in impregnated activated carbon responsible for high rates of H2S removal. In another line of research, we investigated the removal of dyes and different classes of toxins in aqueous solutions, with the use of adsorbent materials such as activated carbons, MOFs and lamellar double hydroxide.
 Carbon pore positioning of H2S molecule in a mixtures of 200 ppm H2S with 40% of CO2
Carbon pore positioning of H2S molecule in a mixtures of 200 ppm H2S with 40% of CO2
Treatment of diseases usually requires or has its efficacy exponentially improved with the use of nanocarriers. These materials provide a stable chemical environment upon which unstable or harmful drugs might be controllably released into the target cells. We investigated the ability of mesoporous Molecular Organic Framework (MOFs) to retain drug molecules through adsorption. Aspects such as biocompatibility and stability of the adsorbent structure are also evaluated.
 Doxorubicin molecules interaction inside MOF MIL100(Fe) pore
Doxorubicin molecules interaction inside MOF MIL100(Fe) pore

